
I imagine a lot of you have done a baking soda and vinegar volcano. You put a little pile of baking soda in a cup and pour vinegar over it and a bubbly eruption ensues. I remember doing this many times as a kid. It’s a good classic activity and kids can have lots of fun doing this one on repeat. You can mix it up with adding food coloring, using pipettes to drip the vinegar, making a mountain out of paper mache, etc. There are also some other fun variations on this activity that you might enjoy. One is inflating balloons!
First, here is what is happening with this activity. You are seeing a couple of chemical reactions occurring when you mix vinegar and baking soda together. The chemical name for vinegar is acetic acid and the chemical name for baking soda is sodium bicarbonate. In addition to their chemical names, we can also write them out with their chemical formulas. Acetic acid is C2H4O2 and sodium bicarbonate is NaHCO3. When these react with each other they mix their elements around in what’s called a double replacement reaction. The sodium (Na) switches places with the hydrogen (H), which you can see in the first line of the picture below. The chemicals have definitely changed, but there isn’t really anything that you would be able to see at this point. It’s what happens next that makes this a fun thing to play with. Each of these new chemicals, now called sodium acetate (NaC2H3O2) and carbonic acid (H2CO3), can change too.
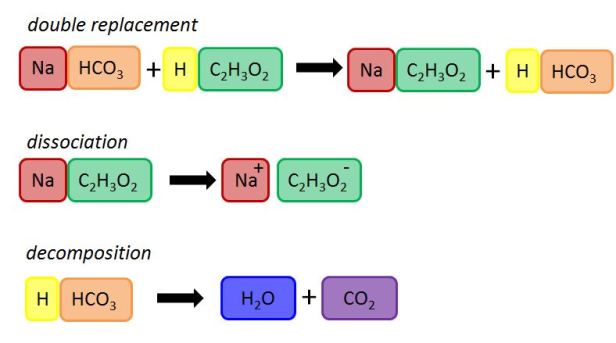
Sodium acetate dissociates in the water which means that the pieces basically fall apart. We can’t really see this either. The carbonic acid, though, does something that we can see. This chemical undergoes a decomposition reaction which means that it decomposes or breaks apart. When that happens, it breaks into water (H2O) and carbon dioxide (CO2). What makes this fun is that carbon dioxide is a gas and you can see it bubble up through the water as it is formed. This is the “eruption” that’s so fun to play with.

So, here is one of the other fun ways to play with it. We can use the gas that is formed to blow up a balloon. Start by pouring some (about 4 Tbs.) vinegar in a bottle. We used beer bottles, but any bottle should work. Then, scoop some baking soda (about 2 tsp.) inside a deflated balloon (a funnel is helpful). Without letting the baking soda spill into the bottle, fit the open end of the balloon over the top of the bottle. Then, tip the balloon up to let the baking soda fall down into the bottle and watch what happens! The gas that is formed is trapped inside the bottle now and as the reaction proceeds the balloon will start to inflate.
Enjoy!